Previous Projects
The mechanisms responsible for oscillations in the rate of CO oxidation at atmospheric pressure is unclear due to the lack of in situ diagnostics in this regime. A number of models have been proposed to describe mechanisms which might be responsible for the observed behavior. We have undertaken studies which attempted to discriminate between the oxidation-reduction and carbon models of this system.
Under stable reaction conditions (where the system temperature and partial pressure of the reactants do not change), the two models predict the same behavior. In order to discriminate between the models, a test was developed in which differences in the transient response of the system to changes in the experimental conditions was measured. In each test, the system was exposed to only one of the two reactants for a long time. The second reactant was then added to the gas stream and the ignition of CO2 formation was monitored.
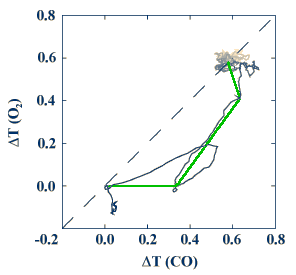
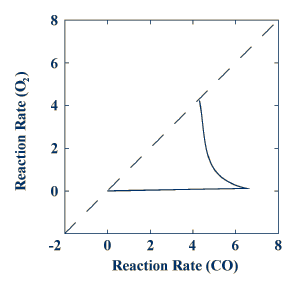
This test was repeated twice - once pretreating with CO and once pretreating with oxygen. The results for the oxidation-reduction model are plotted in the form of a composite graph. In the figure below, the temperature following an oxygen pretreatment is plotted as a function of the temperature following a CO pretreatment. The curve initially follows the horizontal axis, which will be called "CO-like."
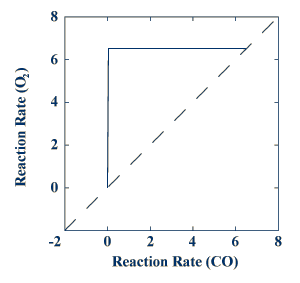
The figure below shows the composite graph for the carbon model. Again, the temperature following an oxygen pretreatment is plotted as a function of the temperature following a CO pretreatment. In this case, the curve initially follows the horizontal axis, and will be called "oxygen-like."
Finally, the figure below shows the composite graph for the experiment. The green line shows behavior of the tests after taking systematic errors into account. Since the curve is initially CO-like, the data support the oxidation-reduction model.